

Household Cleaning Products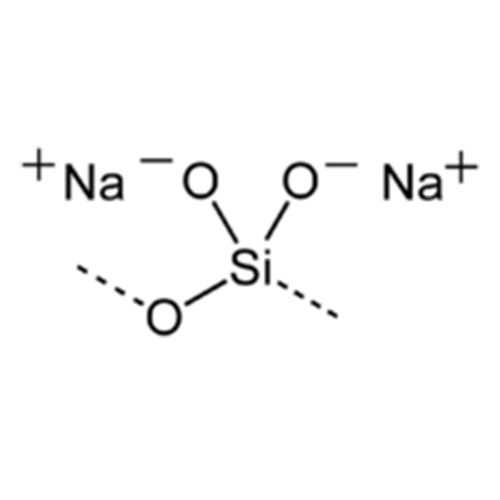
Sodium metasilicate is a member of the sodium compound family, according to the International Programme on Chemical Safety. A very basic compound, sodium metasilicate is used in a variety of industrial and household settings. While it is a cleaning and de-rusting solution, it is highly corrosive and should be handled with care.
Sodium metasilicate is produced by fusing sand with sodium carbonate at very high temperatures, notes the International Programme on Chemical Safety. This produces a clear, white or grayish-white crystal solution. Sodium is a base, and therefore has a high pH level. When interacting with acid, sodium metasilicate will often neutralize that acid.
Routes of exposure
The substance can be absorbed into the body by inhalation of its aerosol and by ingestion.
Inhalation risk
Evaporation at 20°C is negligible; a harmful concentration of airborne particles can, however, be reached quickly when dispersed.
Effects of short-term exposure
The substance is corrosive to the eyes, skin and respiratory tract. Corrosive on ingestion.
Reference: http://bit.ly/1MCajCD
No articles were found